Development of Reference Materials for food allergen analysis
The project successfully produced and characterised the first multi-allergen reference material kit containing five common allergens.
Abbreviations and glossary
| Abbreviation | Meaning of the abbreviation |
|---|---|
| AOAC | Association of Analytical Communities, (formerly Association of Official Analytical Chemists |
| APA | Association of Public Analysts |
| dPCR | Digital PCR |
| AQA | Analytical Quality Assurance |
| EFSA | European Food Safety Authority |
| EHO | Local Authority Environmental Health Officer |
| ELISA | Enzyme-linked Immunosorbent Assay (a type of immunoassay) |
| EuroPrevall | A European and international research programme on the prevalence, cost and basis of food allergy – headed by Prof Clare Mills |
| FSA | Food Standards Agency |
| iFAAM | Integrated approaches to food allergen and allergy management, an international research programme to develop evidence-based management of allergens in food and integrate knowledge derived from their application into food allergy management plans and dietary advice |
| ILSI | International Life Sciences Institute, a non-profit, worldwide organization whose mission is to provide science that improves human health and well-being and safeguards the environment |
| ISO | International Organization for Standardisation |
| JRC | Joint Research Centre (of the European Commission) |
| LGC | Formerly Laboratory of the Government Chemist, now a private company |
| NML | The UK’s National Measurement Laboratory (NML) at LGC is the UK’s designated National Measurement Institute for chemical and bio- measurement |
| MFAN | University of Manchester Food Allergy Network |
| MoniQA | International Association for Monitoring and Quality Assurance in the Total Food Supply Chain, an international and interdisciplinary network working in food research, regulatory bodies and trade. |
| MS | Mass Spectrometry |
| OCL | Official Food Control Laboratory (see below Public Analyst) |
| PA | Public Analyst |
| PAL | Precautionary allergen labelling ('may contain') |
| PCR | Polymerase Chain Reaction, a technique used to amplify DNA sequences so that they can be identified |
| qPCR | Real time-PCR, which uses fluorescent markers on PCR primers to follow the PCR amplification process as it is occurring (for example, in real time) |
| RM | Reference material |
| TSO | Local Authority Trading Standards Officer |
| UKAS | United Kingdom Accreditation Service, licensed to accredit laboratories to the ISO/IEC 17025 standard |
| UoM | University of Manchester |
Executive Summary
This research has now been published as a scientific paper in the Food Chemistry journal, which can be found here.
Reference: Gill Holcombe, Michael J. Walker, Malvinder Singh, Kirstin Gray, Simon Cowen, Stephen L.R. Ellison, Adrian Rogers, Anuradha Balasundaram, Malcolm Burns, E.N. Clare Mills, 2023, Clinically and industrially relevant incurred reference materials to improve analysis of food allergens, milk, egg, almond, hazelnut and walnut, Food Chemistry, 434, 2024, 137391
Food hypersensitivity is a serious and growing problem, of which food allergy is a significant facet with up to about 2 million people in the UK estimated to be affected. In the absence of a currently accepted cure, affected individuals must throughout their life avoid eating foods to which they are allergic. This can be difficult to achieve in practice. Allergens can sometimes find their way into foods unintentionally. Even very small quantities of foods (as little as a hundredth of a gram in the case of some nuts) can cause an unwanted reaction in a person with an allergy to the food.
Analysis of food and food ingredients for allergens is vital to secure safe food for people with allergies, protect the supply chain and support businesses and risk assessors. However, analysis is hampered by lack of reliable reference materials to check and harmonise the proper performance of laboratory tests. Food Standards Agency (FSA) project FS101206 set out to partially address this need. LGC led a consortium with the University of Manchester and Romer Laboratories Ltd which was awarded the project following an open competitive tender.
The consortium members brought unique skills. The UK’s National Measurement Laboratory (NML) at LGC is the UK’s designated National Measurement Institute for chemical and bio- measurement with extensive experience in the production of reference materials to ISO 17034:2016 ‘on the general requirements for the competence of reference material producers’ (previously ISO Guide 34:2009). The University of Manchester (Professor ENC Mills) (UoM) applies molecular science to better understand, diagnose and treat food allergies with experience of running a series of high value EU Framework Programmes on food allergy including EuroPrevall, iFAAM and ThrALL and extensive knowledge of protein chemistry. Professor Mills also runs the Manchester Food Allergy Network (MFAN) bringing together stakeholders in allergen risk assessment and risk management. Romer Laboratories Ltd (Adrian Rogers) (Romer Labs) is a leading global supplier of diagnostic platforms for food safety including food allergens, with ISO/IEC 17025 accredited laboratories in Austria, UK, US and Singapore and a contributor to harmonisation on food allergen measurement.
The project successfully produced and characterised the first multi-allergen reference material kit containing five common allergens (milk, egg, almond, hazelnut, walnut), traceable to the SI (International System of Units). It will help scientists and industry by supporting method development to determine ‘true’ allergen content, and assist laboratories in monitoring the performance of methods on a day-to day basis. This is improving the safety of food products for people with food allergies by helping analysts provide meaningful information for risk assessors.
This report briefly describes the governance of the project, stakeholder engagement to optimise the format and contents of the reference material kit and its preparation and characterisation. More detailed information is available in three peer reviewed publications referenced in the report. We also touch on dissemination of information about the reference materials.
The reference materials have been prepared as a ‘kit’ consisting of
A medium analytical difficulty polyphenol-containing processed food chocolate paste matrix
- (a) devoid of the five allergens, and
- (b) incurred with each allergen at the clinically relevant concentration of 10 mg kg-1 expressed as protein.
The allergen raw materials,
- hens’ egg white powder,
- skimmed cows’ milk powder,
- almond powder (full fat),
- hazelnut powder (partially defatted), and
- walnut powder (partially defatted)
The raw materials are also individually available as well as being included in the kit.
The preparation, gravimetric traceability to the SI, homogeneity and short term (transportation) stability and long term stability of the RM have been demonstrated and are described. Long term stability continues to be monitored. These data together with species characterisation by Sanger sequencing and allergen profiling in the raw materials by a combination of immunoblotting and discovery mass spectrometry demonstrate the suitability of the reference materials. The reference materials, which have been available since early 2020, have been confirmed within the scope of LGC’s ISO 17034 accreditation. Assigned values are compared with independently obtained data from two ELISA platforms and statements of measurement have been published.
The successful conclusion of this project does not, of course, solve all the problems in food allergen analysis and we make recommendations for further work to build on the firm foundations reported herein. These include:
- comparison of data from the reference materials on ELISA platforms other than the two exhibited in the project. Two allergens (almond and walnut) were not funded to be extensively characterised in the project and the reference materials would benefit from further study, including in multiple laboratories.
- although the proteomics of some allergens in the kit have been reported there is scope for further work on the allergen profiles and on value assignment by liquid chromatography –tandem mass spectrometry (LC-MS/MS).
- value assignment for the nut ingredients by Polymerase Chain Reaction (PCR) DNA methods would add to extant data and achieve copy number to mass fraction conversion factors.
- homogeneity data in the reference materials are satisfactory but for hazelnut the data are more dispersed and it is not possible to distinguish the inherent variability of the ELISA from effects perhaps caused by the raw material particle size. The application of digital PCR, (dPCR) which offers much lower variance than ELISA methods and absolute single molecule quantification of DNA species without an external calibration curve, would give more precise information on the homogeneity of the reference materials.
- if dPCR homogeneity data retain the same dispersion driven by the particle size and mixing into the matrix exploration of cryogenic (-80C) milling would be useful. Assessment of the impact on protein structure of these low temperatures would also be interesting.
Food hypersensitivity is an increasing problem for many stakeholders with much effort focused on assessment and management of the risks including risk assessment toolkits (for example, the Allergen Bureau Voluntary Incidental Trace Allergen Labelling VITAL®, the iFAAM consortium and the ILSI-Europe Allergen Quantitative Risk Assessment guidance). These toolkits describe the use of action levels and reference doses to assess the risks. A combination of the estimated eliciting dose of allergenic food, (in milligrams as protein) and the amount of food consumed in a single eating occasion can give a threshold or action level as milligrams (as protein) per kilogram (kg) of food) (mg kg-1 as protein) that would provoke an reaction in a given proportion of the allergic population. The eliciting dose is extrapolated from oral food clinical dose-distribution relationships. The results of analysis can be compared to the thresholds or action levels in risk assessment and risk management. Precautionary allergen labelling, well recognized as sub-optimal, could also be improved via an action level approach. Implementation and regulation depend on the ability to measure allergens properly; yet all current analytical approaches exhibit deficiencies, many of which can be addressed by the proper use of appropriate allergen reference materials (RMs). This report describes a pilot project to:
1) Systematically review allergen analytical targets used in ELISA, PCR and MS to allow the creation of a repository of reliable markers and open access verified allergen sequence sets for the studied allergens;
2) Facilitate a guided stakeholder workshop to achieve consensus on priority allergens, physical format of RMs, incurred concentrations and impact of processing;
3) Prepare a RM kit containing (a) a food matrix shown to be devoid of the target allergens, (b) the food matrix incurred with 5 allergens and (c) the raw materials (the allergenic foods);
4) Disseminate to encourage RM use to achieve tangible improvements in allergen analysis, establish a template of best practice for the future and make recommendations for follow up work to complete a set of RMs for the legislated and priority allergens.
The RM matrix is based on that used for clinical dose-distribution studies and the raw allergen materials have been characterised by proteomics. The matrix and incurred allergens are industrially relevant to processed foods and the allergen concentrations are clinically relevant in the light of eliciting dose studies. The RM kit has been prepared following a well-established process that includes prescribed homogeneity and stability studies and a formal sign-off procedure of the statements of measurement, in accordance with ISO 17034:2016 ‘General requirements for the competence of reference material producers’ (an updated standard originally ISO GUIDE 34:2009). In October 2020 following detailed external assessment the RM kit was confirmed within the NML scope of ISO 17034 accreditation. ISO 17034:2016 covers the production of all reference materials, including certified reference materials. It is intended as part of the general quality assurance procedures of the reference material producer.
LGC formed a consortium which was awarded this project by the FSA following an open competitive tender. The consortium consisted of LGC, the University of Manchester and Romer Laboratories Ltd. The consortium is world leading in (a) the preparation, curation and distribution of RMs in an accredited environment and (b) the characterisation of allergen proteins. Synergy with other concurrent work (for example, by iFAAM, EFSA, ILSI, MoniQA, JRC, and AOAC) has ensured value for money.
This report has been compiled from a review of a broad range of data sources including:
- the scientific literature
- the tender documents
- progress reports and minutes of project meetings
- LGC internal documents and in particular the Material Report[1]
- UoM reports
- Romer Lab reports.
The Food Standards Agency (FSA) is an independent non-ministerial government department working across England, Wales and Northern Ireland to protect public health and consumers' wider interests in food. Food hypersensitivity (food allergy, intolerance and coeliac disease) is a key priority for the FSA. Since 1994, FSA (and its predecessor department) has invested over £20 million in funding over 60 research projects, as part of its Food Allergy and Intolerance Research (FAIR) Programme [2], [3]. The key themes in the current FSA strategy (2022 – 2027) are ‘Food is safe’, ‘Food is what it says it is’ and ‘Food is healthier and more sustainable’[4] Food Hypersensitivity is a key element of the FSA’s Strategy 2022–2027 which notes “We work closely with food businesses to ensure they understand their responsibilities and embed the management of food hypersensitivity in a strong food safety culture. This means consumers can make safer choices about the food they buy.” Hence, providing the means, through appropriate RMs, to improve food allergen analysis aligns with FSA priorities.
The objectives of the present study were as shown in Table 1. The project deliverables and milestones were as shown in Table 2. This report fulfils the final deliverable of the project.
Table 1: Project objectives
| Objective | Objective description |
|---|---|
| 1 | Identification and characterisation of allergenic food ingredients for use in QC and RMs. |
| 2 | Production of the Reference Material kit according to the principles of ISO Guide 34, superseded by ISO 17034:2016 |
| 3 | Characterisation of the incurred reference material |
| 4 | Promote the use of reference materials for method validation and/or calibration with users [dissemination/stakeholder engagement]. |
Table 2: Project deliverables and milestones
*By agreement with FSA certain deliverables target dates were relaxed.
| Deliverable | Target date | Title of deliverable or milestone | Deliverable achieved |
|---|---|---|---|
| D1.1 | 31/02/2018 | Publication regarding allergen targets including verified allergen sequence databases. | Achieved |
| D1.2 | 31/12/2018 | Publication on characterization of exemplar tree nut ingredients (hazelnut, walnut, almond). | Achieved |
| M1.1 | 28/02/2018 | Kick-off meeting and stakeholder workshop. | Achieved |
| M1.2 | 31/03/2018 | Ingredients sourced | Achieved |
| M1.3 | 31/05/2018 | Total protein determination completed | Achieved |
| M2.1 | 31/07/2018 | Food matrix produced | Achieved |
| M2.2 | 31/10/2018, amended to 31/03/2019 | Incurred material produced | Achieved |
| M1.4 | 31/08/2018 | 2D-page characterization completed. | Achieved |
| M3.1 | 31/08/2018, amended to 30/04/2019 | Homogeneity assessment for incurred material completed | Achieved |
| M1.5 | 31/10/2018 | Immunoblotting characterization completed | Achieved |
| M1.6 | 31/12/2018, amended to 31/03/2019 | MS profiling completed | Achieved |
| M3.2 | 31/01/2019, amended to 15/07/2019 | Stability assessment for incurred material completed | Achieved |
| M3.3 | 31/01/2019, amended to 15/06/2019 | Material characterisation completed | Achieved |
| M3.4 | 28/02/2019, amended to 01/08/2019 | Certification meeting | Achieved |
| D3.1 | 31/03/2019, amended to 10/08/2019 | Release of Reference Material Kit | Achieved |
| D4.1 | 31/03/2019, amended to 10/08/2019 |
Promotion of Reference Material Kit |
Achieved and in progress |
| D4.2 | 31/03/2019, amended to 10/08/2019 | Preparation of publication describing the production of the Reference Material. Delivery of final report to FSA. |
Achieved |
The structure and responsibilities of the consortium are outlined in Figure 1. A management committee was formed to oversee the project. The membership of the committee was LGC: Dr Michael Walker (chair) Gill Holcombe, Will Webster (secretariat); University of Manchester: Professor Clare Mills, Dr Chiara Nitride; Romer labs: Adrian Rogers. The committee met face to face and electronically. Six formal meetings were held, generally alternating between LGC Teddington and UoM, Manchester or electronically with further informal telecons as necessary. Internal project meetings took place within LGC on an informal and a formal basis; the formal meetings included a planning meeting for the production of the RM (held 24 May 2018) and a certification meeting (held 30 July 2019). Minutes of all formal meetings are available (on a commercial-in-confidence basis) on request. In addition progress reports were made to FSA on a monthly basis by telecon with Dr Jesus Alvarez-Piñera, latterly including Mr Ross Yarham, and (from 2020) Mrs Bhavna Parmar.
Figure 1: Structure of the delivery team (accessible version)
- FSA
- Project Management Committee: Dr Michael Walker (LGC) Chair, Gill Holcombe (LGC), Professor Clare Mills, Dr Chiara Nitride (UoM), Will Webster (LGC) - Secretariat.
- Gill Holcombe (LGC) - RM Project Leader
- LGC - Dr Steve Ellison RM and Statistics Consultant
- Dr Michael Walker Team Consultant
- Dr Malcolm Burns Principal Scientist
- Dr Thierry Le Goff Commercial Manager
- RM production team
- Molecular Biology Team
- Malvinder Singh Senior Food Analyst (ELISA)
The University of Manchester
- Professor Clare Mills
- Dr Chiara Nitride
- Dr Victoria Lee
- Anuradha Balasundaram
Romer Labs
- Adrian Rogers

4.1 Background
Food hypersensitivity, for example, adverse immunologic (IgE and non-IgE mediated) reactions to food, have resulted in considerable morbidity [5] and reached epidemic proportions in the industrialized world [6], [7] affecting 5 – 6% of young children and 1 -2% of adults [8]. Anaphylaxis, a rapid onset multi-organ system allergic reaction can cause fatalities. The risk of such deaths, though comparatively rare [9], contributes to well-documented detriment to the quality of life for allergic consumers and their families [10], [11], [12].
There are burdens on health care [13], on businesses (food recalls, for example) and regulators [14] and in less developed countries where, owing to poor labelling and awareness, significant challenges may exist. Current reputed cures for food allergies remain experimental and lifelong avoidance of the eliciting food(s) is required. Autoimmune conditions such as coeliac disease and food intolerance [15] also impose significant burdens [16] and strict avoidance of the eliciting foods is usually necessary.
4.2 Food allergen regulation
UK and EU legislation requires food manufacturers to list the ingredients of a food product on its packaging. If one of the 14 regulated allergens (Table 3) (chosen based on the prevalence of adverse reactions amongst consumers in the 27 EU member states) is used as an ingredient or processing aid it must be highlighted within this ingredients list.
The approach for unintended allergens is different: Unintended allergens may enter into the food supply chain at any time during harvest, storage, handling, and/or packaging as consequence of the cross-contact or carry over within processing lines. This represents a food safety hazard. Food business operators should implement a risk assessment in order to establish whether a hazard is likely to occur, and seek to idea; eliminate this risk, or, where this is not feasible, reduce the risk of contamination to acceptable levels, below which no, or only the most sensitive, allergenic subjects might react.
Precautionary Allergen Labelling should be used when there is an unavoidable risk of allergen cross-contact that cannot be sufficiently controlled. Uncertainty around the risk assessing allergen cross-contact has resulted in the untargeted use of Precautionary Advisory Labelling (PAL) - for example, ‘may contain...’. This has caused subsequent confusion of the exact meaning of such warnings amongst consumers [17], [18].
Table 3: Allergens covered by EU labelling law, Annex II Regulation 1169/2011 (UK Retained Regulation (EU) 1169/2011)
| Annex II Entry | Examples |
|---|---|
| Cereals containing gluten and product thereof | Wheat, Rye, Barley, Oats |
| Crustaceans and products thereof | Shrimp/prawn, Crab, Lobster, Crayfish |
| Eggs and products thereof | - |
| Fish and products thereof | - |
| Peanuts and products thereof | - |
| Soybeans and products thereof | - |
| Milk and products thereof | Skimmed milk powder, Cheese etc. |
| Nuts and nut products namely | Almond, Hazelnut, Walnut, Cashew, Pecan, Brazil, Pistachio, Macadamia |
| Celery and products thereof | - |
| Mustard and products thereof | - |
| Sesame seed and products thereof | - |
| Sulphur dioxide/sulphites | - |
| Lupin and products thereof | - |
| Molluscs and products thereof | Mussels, Scallops, Cockles, Oyster, Clam |
There are limited exceptions cited in Annex II of ingredients that do not contain sufficient allergenic protein to elicit a reaction.
4.3 Allergen risk assessment
4.3.1 DG Santé/JRC workshop
In 2016, DG Santé/JRC organised a joint workshop aiming ‘to identify the sequence of steps required for framing the current use of precautionary allergen information and its enforcement across the EU and UK [19]. The major points of the discussion were: (1) legislative and allergy sufferers’ requirements; (2) risk based approaches to allergen management; (3) the role of analysis in enforcing legislation. The outcomes of this discussion will have a significant impact on analytical measurement requirements. As stated in the available feedback report:
- 'Possible agreement on analytical marker(s) and their conversion to a common reporting unit should be encouraged.
- the most appropriate reporting unit for reporting analytical results is mg total allergenic protein ingredient per kg food.
Establishing an expert group to facilitate the progression of all allergenic foods to report in this manner was thought beneficial. This group should consider work done by CEN and other standardisation bodies in the area.
4.3.2 Risk assessment toolkit development
Risk assessment toolkits were developed by the Allergen Bureau Voluntary Incidental Trace Allergen Labelling, VITAL® and the Integrated Approaches to Food Allergen and Allergy Management (iFAAM) consortium to manage food allergen risk [20]. The practical application of quantitative allergen risk assessment has been described by an ILSI-Europe Expert Group [21], [22]. The toolkits use action levels to determine if a corrective action is required. The action level is a combination of the estimated eliciting dose for the different allergens (ED) and the amount of food consumed in a single eating occasion. The eliciting dose is the predicted amount of allergenic food, expressed as mg of total protein that can provoke an allergic reaction in a given percent of the allergic population. It is extrapolated from dose-distribution relationships that are developed using oral food clinical challenge data. Table 4 and equation 1 show the VITAL® 2.0 and VITAL® 3.0 estimated reference doses and the related critical concentrations (mg kg-1 as protein) of the allergen in the food consumed above which an unacceptable risk is to be expected (action level) calculated for a specific amount of food consumed [23].
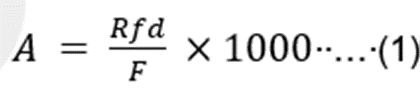
Where:
A: the critical concentration (mg kg-1 as protein) of the allergen in the food consumed
Rfd: the reference dose, in mg as protein as an eliciting dose at a specific population fraction
F : a representative amount of food consumed as g, (x1000 converts to mg kg-1 )
4.3.3 Codex Alimentarius code of practice
In 2020 Codex Alimentarius published a ‘Code of Practice on Food Allergen Management for Business Operators’/ [24]. As part of the ongoing review of the General Standard for the Labelling of Prepackaged Foods Codex also asked the Food and Agriculture Organisation of the United Nations (FAO) and the World Health Organisation (WHO) to look at food allergen risk assessment. A series of four ad hoc Joint FAO/WHO Expert Consultations was set up.
The first reviewed criteria for listing as priority allergens and affirmed the protein based allergens in the Codex list with the addition of sesame. It was recommended that pulses, insects and other foods such as kiwi fruits be included in a “watch list”. The committee also considered analytical capabilities and recommended the development of method performance criteria, more extensive provision of accessible reference materials, better understanding of assay performance and improvements in sampling and curation of samples from originator to laboratory [25]. A full report is available [26].
The second expert consultation discussed ‘thresholds’ for the priority allergens. The experts considered evidence that symptoms up to ED05 doses are mild or moderate. Up to 5 percent of reactions at both ED01 and ED05 could be classed as anaphylaxis, although none were severe. No fatal reactions at or below ED01 and ED05 were reported in the literature. The committee recommended reference doses at ED05 of 1 milligram of the relevant total protein for almond, cashew, pecan, pistachio and walnut, 2 milligrams for peanut, egg, sesame and cows’ milk protein, 3 milligrams for hazelnut protein and for wheat and fish the ED05 reference dose of 5 milligrams. The recommended ED05 for crustacea protein is much higher at 200 mg total protein. A summary [27] and a full report are available[28].
The third consultation discussed precautionary allergen labelling, recognising its difficulties. Their recommendations including that fit for purpose analytical methods are required, including for example matrix-matched assay validation with a limit of quantification at least 3-fold below the action level for the specific food, and reporting in units of milligrams of total protein from the allergenic source per kilogram of food [29].
The fourth consultation considered whether certain highly refined foods and ingredients that are derived from any of the foods on the list of priority allergenic foods could be exempted from mandatory labelling declaration. The consultation aimed to develop a process for the consideration of future such exemptions [30].
Table 4 VITAL® 2.0 and VITAL® 3.0 reference values and calculated action levels to portion size.
Action level (mg kg-1 as protein) based on VITAL 3.0, by way of illustration.
| Allergen | Reference dose (mg of protein): VITAL® 2.0 (V) and/or EACCI (E) *ED01 or #ED05 | Reference dose (mg of protein): VITAL® 3.0 (2019) All ED01 | Examples of amount of food consumed 5g | Examples of amount of food consumed 50g | Examples of amount of food consumed 250g |
|---|---|---|---|---|---|
| Peanut | 0.2* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Cow's milk | 0.1* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Egg | 0.03* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Hazelnut | 0.1* (V,E) | 0.1 | 20 | 2 | 0.4 |
| Soya | 1.0* (V,E) | 0.5 | 100 | 10 | 2.0 |
| Wheat | 1.0# (V,E) | 0.7 | 140 | 14 | 2.8 |
| Cashew | 0.10# (V,E) | 0.05 | 10 | 1.0 | 0.2 |
| Mustard | 0.05# (V,E) | 0.05 | 10 | 1.0 | 0.2 |
| Lupin | 4# (V,E) | 2.6 | 520 | 52 | 10 |
| Sesame seed | 0.2# (V,E) | 0.1 | 20 | 2 | 0.4 |
| Shrimp | 10# (V,E) | 25 | 5000 | 500 | 100 |
| Fish | 0.1# (V,E) | 1.3 | 260 | 26 | 5 |
Note 1: The amounts shown here are only for illustration purposes. For guidance on how to estimate the amount of food consumed see ILSI-Europe, Practical Guidance on the Application of Food Allergen Quantitative Risk Assessment, Section 5.3: Guidance on food intake data for allergen risk assessment.
Note 2: In the last three columns of Table 4 only the appropriate number of significant figures have been retained.
4.4 Food allergen analysis
Reliable analytical measurement of allergens is required for many reasons [32] including to support the validation and verification of risk assessment plans in food industrial plants. This will allow food business operators to obtain quantitative values, expressed as mg of total allergenic protein per kg of food product, to be used in the process of decision-making.
4.4.1 Enzyme-Linked Immunosorbent Assay (ELISA)
The food industry and enforcement scientists (Public Analysts) largely employ Enzyme-Linked Immunosorbent Assay (ELISA) for routine assessment of food allergen concentrations. The technique is relatively easy to use, specific, not overly time consuming, and exhibits suitably low limits of detection and quantification for many allergens. However there are limitations. The assay can detect only one allergen per test and responses can be affected by nonspecific antibody reactivity and the non-reversible modification of the targeted protein(s) during food processing can mask the epitope to the antibody hence the risk of false positive or negative results is high. The lack of standardization can generate conflicting results among ELISA kit manufacturers. In proficiency testing multimodal datasets for allergen ELISAs are common and different assigned values have to be generated for the different kits used. Cross reactivity’s to foods other than the intended target have been reported for what were assumed to be specific ELISAs.
4.4.2 Polymerase Chain Reaction (PCR)
Polymerase Chain Reaction (PCR) assays are also applied in allergen analysis. They do not directly measure the hazard (protein), and are probative of the source species DNA rather than the allergen protein. They are especially valuable for those foods for which no ELISA based test is available and often have advantages in terms of sensitivity and specificity. In addition, as an analyte DNA is generally more stable than proteins although DNA degradation does occur by extremes of, for example, temperature and pH. PCR is essentially qualitative at present. Quantification based on DNA copy number can be derived from cycle thresholds but requires reference materials to construct a calibration curve (although digital PCR may circumvent this difficulty). However at present it is not easy to convert a quantification based on copy number to a weight/weight basis.
4.4.3 Mass Spectrometry (MS)
Mass spectrometry (MS) platforms are becoming the gold standard technologies for protein characterisation and quantification. Shotgun and targeted protein identification and quantification by mass spectrometry have been successfully applied to the analysis of allergens in processed food matrices.
Mass spectrometry allows a bottom-up analysis using harsher extraction protocols than can be used with ELISA (where it is essential to preserve the intact and native structure of the protein). Under MS, crude extracted protein can be broken down into its constituent peptides using enzymatic digestion.
The shotgun approach provides information on protein/peptide structure and on chemical/thermal modifications occurring during food processing. The targeted approach, by monitoring specific peptide markers as surrogates of given allergenic proteins, is capable of measuring many analytes in parallel, resolving very complex proteomes and dealing with protein abundances different by four to five orders of magnitude, while maintaining high mass accuracy. However, the technique requires a high level of expertise and costly equipment. More importantly, the preparation of the analytes for mass spectrometry analysis, including extraction and digestion of the proteins, remains challenging.
4.4.4 Overview
In summary, current industrial practice is heavily reliant on ELISA, and the method has brought many benefits in allergen risk management. However it exhibits some deficiencies which may jeopardise present and future risk management strategies. Alternative methods of allergen analysis are promising, but are also not without their challenges. The promise of MS or PCR may be lost if underpinning work suggested herein is not carried out. Structural changes in the target molecules induced by food processing or sample extraction may prevent detection by all methods.
5.1 Background
What can be done to address the limitations of all the available approaches to food allergen analysis? One solution is the availability of well characterised calibrants and reference materials (RM) to allow comparability and harmonisation of measurements obtained using diverse analytical methods and across different laboratories. More importantly, the production of reference materials which represent a real food product would help analysts to develop methods capable of providing food business operators with meaningful information that can be easily used for implementing risk assessment plans.
Work undertaken by the Joint Research Centre (JRC) has attempted to develop a candidate RM in relation to ELISA measurements of gluten. Gluten represents a challenging analyte as it is not clearly defined and there is no agreed common marker or conversion factor. The results of the work were mixed. On the one hand, the use of the gluten reference material as a calibrant helped to reduce within-kits and between-assay variability somewhat. On the other hand, when different kits target diverse fractions of the gluten molecule, this naturally leads to different antibody selectivity which was not improved by the use of a common calibrant [33]. Such data emphasises the need for development of internationally recognised sets of analytical targets and allergen reference materials to improve the reliability of allergen analysis.
Reference material (RM) and Certified Reference Material (CRM) are well defined terms within an associated international infrastructure. Food allergen RMs should comply with this infrastructure. Reference materials produced by National Measurement Institutes such as LGC exhibit the highest standards. Taylor et al [34]. were probably among the first to describe with examples the preparation of naturally incurred standards as allergenic food residues incorporated into various representative food matrices and then processed in a manner similar to ‘real-world’ food processing. This approach has stood the test of time in this and other contexts. Thus it is routine to produce very useful RMs of food in food (meat species for example) and LGC are long accustomed to analysing for food in food to appraise QUID (Quantitative Ingredients Declaration) – required by labelling law for certain foods.
5.2 Commercially available materials
5.2.1 Reference materials
The consortium investigated the reference materials already available on the market. They take two different forms: raw ingredients and incurred processed food matrices. The analysis included the major developers and producers of reference materials with Joint Research Centre (JRC), National Institute of Standards and Technology (NIST), LGC, and the National Metrology Institute of Japan (NMIJ). It is noteworthy that most were not originally intended as allergen protein reference materials.
5.2.2 Raw ingredients
Several milk powder reference materials are certified for the content of trace elements, organic pollutants, veterinary residues, natural toxins, and vitamins. JRC and NIST produce milk reference material as skimmed milk powder (BCR – 685) and whole milk powder (NIST – SRM 1549a) that can be used for measuring the nutritional values of the food product. The materials are certified for the crude protein content determined by Kjeldahl analysis. A factor of 6.38 was used to convert nitrogen results to protein.
A spray dried whole egg for allergen detection, produced without additives or stabilizers, is available from NIST (NIST – SRM 8445). The approximate content of protein was measured using a Leco protein analyser (Dumas). NIST also produces a whole egg powder for measuring the nutritional values of the food product (NIST – SRM 1845a). Nitrogen was determined by collaborating laboratories using Kjeldahl, thermal conductivity, and combustion (LECO). A factor of 6.25 was used to convert nitrogen results to protein.
A dry whole egg powder reference material (EGGS-1) is also available from the National Research Council Canada (NRC), where the protein concentration was determined using a factor of 6.25 to convert nitrogen in protein.
The JRC made available a material for peanut analysis including 6 vials containing peanuts prepared at different roasting temperatures and times (IRMM – 481). The kit is not considered a reference material since it was not tested for homogeneity and stability.
NIST produced a peanut butter standard reference material certified for the content of proteins (NIST – SRM 2387). A factor of 5.46 was used to convert nitrogen results to protein.
LGC has made available a mechanically defatted light roasted peanut flour in 5 g vials under Argon. (LGCQC1020) which is from the same source and batch of peanut flour used for recent protein detection studies. Values are provided for nitrogen and water content and the material will prove useful for allergen protein measurement.
5.2.3 Incurred processed food matrices
At the beginning of 2017, the MoniQA association made available the first validated reference material for food allergen analysis. They manufactured milk powder cookies at two agreed concentrations, 3.5 (LOW-MQA 102016) and 35 (HIGH-MQA 082016) mg of milk protein per kg of cookie. The materials are sold alongside the dry skimmed milk powder (SMP-MQA 092014) and a blank cookie (BLANK-MQA 082015). In the future, the production of egg and soy-based reference materials is expected.
The FAPAS (Food Analysis Performance Assessment Scheme) produced a cake mix reference material containing gluten, egg, and milk (FCAL7-PRO10RM). Differently from the MoniQA material, egg and milk were added as a known amount of total ingredient to a commercial cake mix manufactured to be gluten-free, egg-free and milk-free. The amount of egg and milk proteins is not traceable. Moreover, the effect of processing on the detection of the allergens is not taken into consideration. The material is currently not available. FAPAS produces two quality control materials: cooked biscuits containing hazelnut and peanut (T27171QC) and chocolate containing hazelnut (FCAL8-CON2QC). For all the products described, the protein concentration assignment was done by consensus assessment based on ELISA analysis.
LGC have made available a quality control set based on chocolate dessert. Each set contains a blank unit and a unit with 10 mg kg-1 gravimetrically incurred peanut protein (LGCQC101-KT). Within the iFAAM project, the material was used as a ring test sample for ELISA and Mass spectrometry analysis.
Synergy with other work (for example, by iFAAM, EFSA, ILSI , MoniQA, JRC, and AOAC) has benefited this project and the consortium was well placed to engage with these stakeholders. The University of Manchester Food Allergy Network, MFAN, which meets alternately at University of Manchester and LGC was also well placed to engage with ELISA kit manufacturers, retailers, analytical service laboratories, regulators and food manufacturers. LGC is actively engaged with Public Analysts, providing the Association of Public Analysts Training Officer role. Particular synergy was established with the EFSA-funded project ThrALL, which aims inter alia to improve the detection and quantification of food allergens. A joint project meeting of ThrALL [34], [35] and FS 101206 project members was held on 22 March 2018. During this meeting it was evident that the activity would not overlap with the outputs of the ILSI working group in which the JRC was involved. Moreover, complementary work ensuing from ThrALL included a study on two highly processed matrices, chocolate bars and broth powder. These were incurred with six allergenic ingredients (egg, milk, peanut, soy, hazelnut and almond) at 2, 4, 10 and 40 mg total allergenic protein/kg food matrix using a pilot-scale food manufacturing plant. All the allergens tested were found to be stable in the incurred matrices for at least 30 months demonstrating they are suitable for method development [35].
The first (and so far the only) reference food allergen method traceable to the SI to enable the comparability of food allergen measurement results expressed in a decision-relevant manner was published in 2020 [36] based on work by Nitride et al. 2019 [37] A workflow and stoichiometric calculations were demonstrated. Challenges included optimal extraction of marker proteins, complete digestion and equimolar release of peptides and the use of conversion factors to translate the amount of measured proteins into allergenic food. Importantly, the combined uncertainty of the final result was reported. This was followed by an interlaboratory comparison that indicated further harmonisation is required quantitatively to determine potentially allergenic constituents in food. [38] The assignment of the reference value for the interlaboratory comparison has been described noting that the proper application of isotope dilution mass spectrometry (IDMS) provides the shortest traceability to SI units and reference values with the lowest uncertainties. [39] A certified reference material for milk protein is expected from this work.
7.1 D1.1 Publication - allergen targets including verified allergen sequences databases
A systematic review of analytical targets used in ELISA, PCR and MS methods was carried out ensuring synergies, in particular, with iFAAM and ThrALL. A joint project meeting was held on the 22 March 2018 between ThrALL and FS 101206 project partnerships. Having well defined allergen sequence sets is crucial to support effective allergenic food biomarker identification and characterisation. The curated allergen sequences were analysed to understand how the protein and peptide markers identified in part 1 relate and whether they cover major isoforms and how factors such as post translational modifications and processing-induced modifications, such as the Maillard reaction, may impact on the generation and detection of allergen peptide biomarkers by mass spectrometry. The results of the study were published in 2020 covering four of the five allergens represented in the RM kit. Financial support from the UK Food Standard Agency (FSA), Contract FS101206 Development of Quality Control Materials for Food Allergen Analysis was acknowledged. The authors, title and abstract are given in Appendix 2, paper D1.1.
7.2 D1.2 Publication on characterization of exemplar tree nut ingredients
Although the RM kit is fully characterised from the point of view of SI traceability, homogeneity, stability and assigned values it is also important to have regard to the allergen proteins represented by the RM. Thus, of the five allergens in the RM kit, three, hazelnut, walnut and almond ingredients were studied. The results of the study are due to be published. Financial support from the UK Food Standard Agency (FSA), Contract FS101206 Development of Quality Control Materials for Food Allergen Analysis is acknowledged. The authors, title and abstract are given in Appendix 2, Paper D1.2
7.3 M1.1 Kick-off meeting and stakeholder workshop
7.3.1 Kick-off meeting
A kick-off meeting was held at LGC Teddington on 23 February 2018 with Gill Holcombe, Thierry le Goff, Gavin Nixon, Malvinder Singh, Michael Walker, Will Webster (LGC); Jesus Alvarez-Pinera† (FSA); Anuradha Balasundaram*, Victoria Lee*, Clare Mills* (UoM); Adrian Rogers* (Romer),
† via telephone; * via Skype.
The Agenda was as follows:
- Introduction to LGC – Gill Holcombe (LGC)
- the need for allergen reference materials – Michael Walker (LGC)
- Project Overview – Michael Walker
- making allergen reference materials – Gill Holcombe
- UoM deliverables – Clare Mills, Anuradha Balasundaram, Victoria Lee (UoM)
- LGC deliverables – Gill Holcombe
The stakeholder meeting and consultation were discussed, followed by sourcing of ingredients for the RM and homogeneity and stability studies. FSA outlined their reporting requirements and project management was discussed. Minutes of the meeting were circulated to all attendees.
7.3.2 Stakeholder workshop
An all-day stakeholder workshop was held on 23 March 2018 at the University of Manchester Institute of Biotechnology in the form of a Food Allergy Network (MFAN) Stakeholder Meeting on the topic’ ‘Reference materials and reference methods for food allergen analysis’. The meeting was held in conjunction with the EFSA funded ThRAll project (GP/EFSA/AFSC, 2017/03) with a substantial portion of the discussions devoted to project FS101206).
Minutes of the meeting were forwarded to FSA. The discussion sessions were used to gather stakeholder views on allergen RMs which included:
- synergy rather than duplication with ThrALL and MoniQA
- the utility of RMs except when ELISA platforms measure entirely different epitopes
- priority allergens were milk, gluten, egg, soy, peanuts, nuts and fish
- the form used in the RM must be representative of an industrial-scale allergen food and would also want matrix-matched calibrator
- there was a discussion regarding the use of whole egg or egg white powder
- the matrix should be simple or of medium analytical difficulty, problematic matrices included takeaway food, baked goods, other processed foods in complex recipes, cooked and raw materials and spices
- incurred concentrations should be between the LoQs of test kits, and levels of clinical effects
- conversion factors to mg kg-1 of the relevant allergen protein are important
- any usable RM should be stable for around 3 to 5 years
The agenda is shown below, in two parts, morning and afternoon sessions.
| Morning | Event | Speaker |
|---|---|---|
| 9 to 9:30am | Registration and coffee | - |
| 9:30am to 9:35am | Welcome | Clare Mills, The University of Manchester UK |
| 9:35am to 9:55am | Detection and quantification of allergens in foods and minimum eliciting doses in food allergic individuals (EFSA-project ThRAll) | Clare Mills, The University of Manchester UK |
| 9:55am to 10:15am | Towards reference materials for food allergen analysis (FSA project FS101206) | Michael Walker, LGC, UK |
| 10:15am to 10:35am | Reference materials - the MoniQA experience | Roland Poms, MoniQA, AT |
| 10:35am to 11am | Allergenic ingredient and incurred food matrices | Christof van Poucke, ILVO, BE |
| 11am to 11:30am | Coffee break | - |
| 11:30 am to 1pm | What makes a usable reference material? Short presentations from Gill Holcombe (LGC) Structured discussion on reference materials What is needed? In what format? How should they be used? |
Facilitator Michael Walker (LGC) and representatives of Contracting analysis Laboratory Research Laboratory Public Analyst Allergen testing commissioning organisation |
- need for advice on how to use an allergenic ingredient for use in preparation of in-house QC materials
- unit size was discussed; for “raw” ingredient about 1 g would be an acceptable if spiking into a matrix. For the incurred matrix a larger number of smaller units is preferable, or multiple units per kit would be beneficial.
7.3.3 Further stakeholder consultation
Following the stakeholder meeting a further emailed consultation exercise was carried out on the format and content of the RM. Emails were sent to 41 stakeholders (Figure 2) outlining the background to project FS101206, information on the proposed allergen RM kit, relevant reference doses and the statement that “We intend to incur each allergen at 10 mg kg-1as allergen protein. Do you agree? Or can you suggest an alternative concentration?”
The afternoon agenda session agenda was as follows:
| Afternoon | Event | Speaker |
|---|---|---|
| 1pm to 2pm | Lunch | - |
| 2pm to 2:20pm | Can mass spectrometry provide the reference method for allergen analysis | Gavin O'Connor, JRC, BE |
| 2:20pm to 2:40pm | Standard methods for allergen analysis - AOAC perspectives | Linda Monaci, ISPA, IT |
| 2:40pm to 3pm | The perspective of a routine analytical laboratory | Nathalie Gillard, CER, BE |
| 3pm to 3:30pm | Coffee Break | - |
| 3:30pm to 4pm | ELISA and PCR analysis - how to integrate with MS methods |
Olivier Tranquet, INRA, FR Marc de Loose, ILVO, BE |
| 4pm to 4:30pm | Integrating ELISA and MS analysis of peanut | Vicki Lee, The University of Manchester, UK |
| 4:30pm to 5pm | Topics for future meetings | All |
| 5pm | Meeting close | - |
There were 13 responses (to 22.11.2018) which can be summarized (Figure 3) as follows:
- most (9/13 = 69%) broadly agreed 10 mg kg-1 as allergen protein (10 mg kg-1 AP)
- some noted 10 mg kg-1AP was high but commented RM could be diluted
- some preferred 5 mg kg-1 AP, or even lower for egg
It was pointed out that with ELISA recoveries of typically 30% – 50%, 10 mg kg-1 AP incurred would probably result in 3 – 5 mg kg-1 AP on typical ELISA calibration curves and it was agreed that would be suitable for analysis by most ELISA kits
Figure 2: Stakeholder groups consulted on the RM format and content
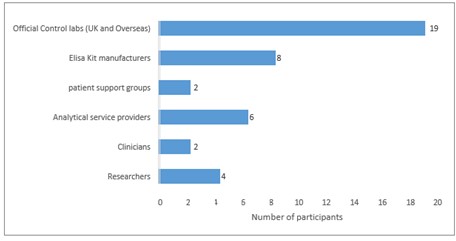
Figure 3: Stakeholder responses summary RM format and content

Based on the stakeholder feedback and initially aiming at European and North American populations it was decided that a RM kit should be prepared aimed to be of medium analytical difficulty and complementary to other RMs available (for example, from MoniQA). The RM kit should contain the following:
A food matrix based on the EuroPrevall chocolate dessert mix paste gravimetrically incurred with 5 allergens at clinically and industrially relevant concentrations, (10 mg kg-1 expressed as protein, for example, each of five allergenic foods was added such that the incurred material contains 10 mg kg-1 expressed as protein of each)
The food matrix devoid of the target allergens, and the allergen raw materials which should be
- hens’ egg white powder,
- skimmed cows’ milk powder,
- almond powder
- hazelnut powder, and
- walnut powder.
7.4 M1.2 Ingredients sourced
The ingredients for the RM kit were sourced as described in a published paper (see 8.20). Data collected have been archived for retention in accordance with LGC document control policies.
7.5 M1.3 Water and total protein determination
The water and total protein contents of the allergenic ingredients for the RM kit were determined as described in a published paper (see 7.17). The datasets from the determinations have been archived for retention in accordance with LGC document control policies.
7.6 M2.1 Food matrix produced
The food matrix for the RM kit was determined as described in a published paper (see 7.17). The written procedures, witnessed key steps and data collected have been archived for retention in accordance with LGC document control policies.
7.7 M2.2 Incurred material produced
The incurred material for the RM kit was determined as described in a published paper (see 8.20). The written procedures, witnessed key steps and data collected have been archived for retention in accordance with LGC document control policies.
7.8 M1.4 2D-page characterization completed.
See 8.2 D1.2, publication, Adaba et al., 20XX, Characterisation of hazelnut, walnut and almond ingredient for use as reference materials for allergen analysis.
7.9 M3.1 Homogeneity assessment for incurred material
The homogeneity of the elements of the RM kit was determined as described in published paper (see 7.17). The datasets from the determinations have been archived for retention in accordance with LGC document control policies.
7.10 M1.5 Immunoblotting characterization
See 7.2 D1.2, publication, Adaba et al., 20xx, Characterisation of hazelnut, walnut and almond ingredient for use as reference materials for allergen analysis.
7.11 M1.6 Mass spectrometry, MS, profiling
See 7.2 D1.2, publication, Adaba et al., 20xx, Characterisation of hazelnut, walnut and almond ingredient for use as reference materials for allergen analysis.
7.12 M3.2 Stability assessment for incurred material
The stability of the elements of the RM kit was determined as described in published paper (see 7.17). The datasets from the determinations have been archived for retention in accordance with LGC document control policies.
7.13 M3.3 Material characterisation completed
The stability of the elements of the RM kit was determined as described in published paper (see 7.17) and Adaba et al., 20xx, Characterisation of hazelnut, walnut and almond ingredient for use as reference materials for allergen analysis, read 7.2.
7.14 M3.4 Certification meeting
A certification meeting was held at LGC on 30 July 2019.
Present: Stephen Ellison (PS/Reference materials, Chair), Gill Holcombe (Hd/RM production, RM project leader), Natasha Heath (Quality manager), Kirstin Gray (Food analyst), Malvinder Singh (Food analyst), Simon Cowen (Statistics), Laura Carrick-White (RM Production team), Nagina Junaid (RM Production team), Michael Walker (Project management committee chair; Hd/Office of the Government Chemist).
Present for the first half hour: Cailean Clarkson (Team leader, purity analysis team), Michael Tong (analyst, purity analysis team).
Information was presented by Gill Holcombe on the
- preparation of the material,
- homogeneity and stability data and their statistical evaluation
- assigned values, uncertainty and metrological traceability
- intended use and instructions for use.
All aspects of the RM were critically reviewed and a series of actions agreed.
7.15 D3.1 Release of Reference Material
On satisfactory completion of the actions agreed at the certification meeting the RM was confirmed as suitable for release for sale and statements of measurement were drawn up and agreed. The RM kit and associated RMs (raw materials) were made available in February 2020 with a press release, pre-agreed with FSA on 10 February 2020. The descriptions of the RM are as follows and the statements of measurement are available open access on the LGC website (links below).
7.15.1 Allergen kit – Milk, Egg, Almond, Hazelnut and Walnut, Reference Material LGC746-KT
Kit contents, each kit contains:
One vial of each of the following raw materials, each containing ~1 g under Argon, each characterised for total nitrogen (Dumas) and moisture (Karl-Fisher):
- LGC7421 Skimmed milk powder
- GC7422 Egg white powder
- LGC7424 Almond powder
- LGC7425 Hazelnut powder – partially defatted
- LGC7426 Walnut powder – partially defatted
Five bottles, each containing ~5 g, of:
- LGC7461 Chocolate paste – no added allergenic ingredients
- LGC7462 Chocolate paste with each of the above added allergenic ingredients gravimetrically incurred by serial dilution of a high-level paste at 10 mg kg-1 (expressed as protein calculated on total nitrogen in the original raw material).
The Statement of Measurement contains:
- for LGC7421, LGC7422, LGC7424, LGC7425 and LGC7426 a table of assessed values for total nitrogen (Dumas) and moisture (Karl-Fisher) with associated uncertainty, the half-width of the expanded uncertainty interval calculated using a coverage factor (k) of 2 which gives a level of confidence of approximately 95 %.
- for LGC7421, LGC7422, LGC7424, LGC7425 and LGC7426 a table of calculated values for protein content with associated uncertainty and the relevant nitrogen factor. The calculated values table has been included for information. Whilst it is known that the nitrogen factor will vary according to the source of the ingredient, no allowance associated with the factor uncertainty (which is known to be non-zero) has been included in the calculation of the protein content and its uncertainty, nor has this source of uncertainty been included in the uncertainty for the assessed and indicative values for allergen proteins. Users may wish to include their own estimates of this source of uncertainty.
- for LGC7461 a table of assessed values for milk protein (<0.05 mg kg-1), egg white protein (<0.05 mg kg-1) and hazelnut protein for LGC7462 a table of assessed values for milk protein, egg white protein, and indicative values for hazelnut protein, almond protein and walnut protein based on the gravimetric preparation data with their associated uncertainty, the half-width of the expanded uncertainty interval calculated using a coverage factor (k) of 2 which gives a level of confidence of approximately 95 %, except for the hazelnut protein data where a tolerance interval calculated from log-transformed data is reported owing to an observed larger between-unit standard deviation.
- a table of results for LGC7462 obtained by analysis using two different commercial ELISA kits. In most cases the mean result measured is lower than the prepared concentration, which is not unexpected. For example, for ELISAs calibrated using ’whole’ hazelnuts (not defatted), low recovery from partially defatted nut powders is well known, and may be related to the impact of lipid removal on proteins. The table shows the study design and range of individual results obtained; however these data should be used with caution, as the range is likely to be less where a small number of units are examined, and a user may obtain results outside this range in their laboratory.
- a table showing material sourcing and preparation.
- a table of conversion factors from total nitrogen to casein, hazelnut, almond and walnut.
- statements of homogeneity and stability assessments with brief comments on the study designs and outcomes.
- brief descriptions of analytical methods (nitrogen, water, DNA sequencing), and proteomics.
- a description of the intended use and instructions for use.
- a statement of the metrological traceability.
7.15.2 Allergen ingredient reference materials -
Statements of measurement are also freely available for:
- Skimmed milk powder LGC7421
- Egg white powder LGC7422
- Almond powder LGC7424
- Hazelnut powder – partially defatted LGC7425
- Walnut powder – partially defatted LGC7426
Each gives details of the nitrogen and water content of the materials with associated uncertainty and methods of analysis, the material sourcing and preparation, the homogeneity and stability assessments, the intended use, metrological traceability, accreditation, instructions for use, storage conditions and shelf life. DNA sequencing information is given for the tree nuts.
7.15.3 ISO 17034 accreditation
ISO 17034:2016, ‘General requirements for the competence of reference material producers’, covers the production of all reference materials, including certified reference materials. It is intended as part of the general quality assurance procedures of the reference material producer. This International Standard supersedes ISO Guide 34:2009 and is aligned with the relevant requirements of ISO/IEC 17025. Reference material producers that comply with this International Standard will also operate generally in accordance with the principles of ISO 9001:2015 ‘Quality management systems, Requirements’. Accreditation to ISO 17034 may only be awarded after the RM is produced, so that its production, evaluation and certification may be audited. Following such an audit in 2020, the allergen RMs were confirmed within the scope of LGC’s ISO 17034 accreditation.
7.16 D4.1 Promotion of Reference Material kit
Press releases for the release of the RM kit and award of ISO 17034 were issued on 10th February 2020 and 16th October 2020 respectively. Each was advised to FSA prior to issue. The LGC website is being used to promote the RM kit which has been flagged on social media. A story on the RM was carried by ‘Food Manufacture’ on 10 February 2020, and on the University of Manchester and Romer Labs websites.
The release of the RM kit was publicised at the following conferences:
- FOOD ALLERGY - Human, Analytical & Regulatory Implications hosted by Institute for Global Food Security, Queen’s University Belfast, Monday 21st October 2019, Riddel Hall, Belfast, Michael Walker
- NIST Food Safety Workshop, Gaithersburg, Maryland, USA, October 28 – 31 2019, Gill Holcombe and Michael Walker.
- MoniQA, Food Fraud Prevention and Effective Food Allergen Management, Rockville, Maryland, USA, 30 October – 1 November 2019, Gill Holcombe and Michael Walker.
- FAAM virtual conference on 16 and 17 October 2020. This is a major clinical and scientific food allergy biennial conference in the European Academy of Allergy & Clinical Immunology (EAACI) events calendar. It was due to be held in Manchester with Professor Clare Mills as chair of the multinational organising committee however COVID-19 forced a virtual conference which attracted over 800 delegates. Michael Walker was an invited speaker, giving a talk entitled "The pitfalls of food testing and electronic allergen information provision" which included reference to the RM kit. This talk was also selected to be included in an EAACI medical training module.
7.17 D4.2 Publication, production of Reference Materials and FSA final report
A paper on the production and characterisation of the RM kit has been prepared for publication. Financial support from the UK Food Standard Agency (FSA), Contract FS101206 Development of Quality Control Materials for Food Allergen Analysis is acknowledged. Authors, title and abstract are given in Annex 2. paper D4.2.
This report constitutes the Final report to FSA.
An internal 229-page LGC report [1] contains the complete dataset on the allergen reference materials and is available on request for RM users and at the discretion of the Head of RM Production.
8.1 Background
Food allergy is an increasing problem for those affected, their families or carers, the food industry and for regulators [40], [41]. The food supply chain is vulnerable to fraud involving food allergens and in the catering sector instances of poor compliance with basic allergen management have occurred with high media salience [44], [45], [46], [47]. There is the risk of fatalities and severe reputational damage to the food industry. Precautionary allergen labelling is widely regarded as suboptimal and in need of standardisation [48].
Many facets are being pursued to ameliorate the difficulties including better food labelling and the concept of thresholds of elicitation of allergy symptoms as risk management tools [46]. These efforts depend to a high degree on the ability reliably to detect and quantify food allergens; yet all current analytical approaches exhibit deficiencies that jeopardise accurate results being produced particularly in terms of the risks of false positive and false negative reporting. Three distinct but interrelated areas of analytical work have been recommended [22] to address the substantial gaps identified:
1. a coordinated international programme for the production of properly characterised clinically relevant reference materials and calibrants for food allergen analysis
2. an international programme to widen the scope of proteomics and genomics bioinformatics for the genera containing the major allergens to address problems in ELISA, MS and DNA methods.
3. the initiation of a coordinated international programme leading to reference methods for allergen proteins that provide results traceable to the SI.
Recommendation 1 has attracted much support [47], [48]; the work reported herein is a step in addressing recommendation 1 and in part recommendation 2. Confidence in analytical results is related to the availability of reference materials (RMs), which play an essential role in method calibration and validation, measurement uncertainty assessment, proficiency testing and quality assurance in order to guarantee accuracy and reliability of the analytical data, as well as comparability of results from different laboratories. Unlike reference materials for single analyte molecule or well-defined classes of compounds, the preparation and certification of RMs for food allergens are very challenging [43].
The consortium that carried out this study has previous experience [49], [50], in the preparation and utilisation of incurred materials that have proved useful in food allergen analysis. However given the constraints of the study it was important to consult the analytical community most concerned with food allergen analysis prior to planning and executing the preparation of the RM described herein, described in detail in section 7.3.
The consultation confirmed the utility of the medium difficulty industrially relevant Europrevall cold swelling chocolate flavoured vehicle as a relevant matrix in which to incur priority allergens. It also confirmed the priority allergens as hens’ egg white powder, skimmed cows’ milk powder, and the nuts almond, hazelnut and walnut. All of these are contained in the food allergens subject to specific labelling requirements in European law [51]. Equally importantly it emerged that as well as an incurred matrix, a blank matrix and the food allergen commodities, as they appear in the food supply chain, were needed by the analytical community. The latter address a need for spike recovery experiments in matrices such as catering sector meals for which it is unlikely that dedicated RMs will appear owing to the diversity of the potential matrices.
Thus the RM has been prepared as a kit containing allergens chosen after consulting stakeholders, aimed to be of medium analytical difficulty and to be complementary to other RMs available (for example, from MoniQA).
8.2 Raw materials
The raw materials have been carefully sourced and characterised by proteomics (UoM, led by Professor Clare Mills), and at LGC by PCR and for Dumas N and Karl Fisher water, Figure 4.
Figure 4: Characterisation of the raw material allergens

8.3 The matrix
The matrix is based on a food vehicle designed to blind the participants in oral food challenges to increasing doses of allergens to establish dose-distribution curves of allergic reactions. In general, the ingredients of the RM and the incurred allergens are from the same or similar sources as the foods used in the EuroPrevall studies that gave rise to reference doses for several allergens, hence the clinical relevance of the material. The approximate composition of the paste is shown in Figure 5. It was checked for workability - it is solid at room temperature and a viscous liquid at 37 C, the temperature at which mixing of the incurred allergens occurs and at which it is dispensed. It has been shown to be stable owing to its low water activity and devoid of the allergens intended to be incurred into it. There is no need to rehydrate this RM and it can be used directly from the bottle.
Figure 5 Overview of the RM kit

8.4 The incurred matrix
The allergen concentrations in the incurred material were prepared by weighing out appropriate amounts of each of the raw materials based on their total protein content. A high-level concentration was prepared to a checked written protocol and the final incurred concentration was arrived at by gravimetric serial dilution of the original preparation. Homogeneity and stability studies were carried out to the principles of ISO 17034 2016 (General requirements for the competence of reference material producers). From these, and separate ELISA assays within LGC, information can be given in the statement of measurement on the likely concentrations expected to be arrived at by laboratories using ELISAs from Romer and R-Biopharm.
8.5 Homogeneity
The raw materials and the blank matrix were examined for homogeneity and found to be fit for purpose in this respect. The incurred matrix was similarly studied and found to be sufficiently homogenous for the intended purpose with regard to its incurred hens’ egg white protein and skimmed-cows’-milk protein. For hazelnut protein the homogeneity of the incurred matrix was challenging however a statistical model, with 95%/95% tolerance intervals calculated assuming (a) normality and (b) lognormality was developed that addresses the needs of potential users of the RM. Although funding was not available to assess the homogeneity of almond and walnut protein it is reasonable to assume a similar approach will be required.
In part the challenges to homogeneity studies arise from the available precision of current methods of allergen analysis. However, high lipid content materials such as nuts also present challenges in milling to a required low enough particle size to ensure adequate mixing. This is a topic for further work.
8.6 Stability
Not unexpectedly stability of the incurred material began to falter at 60C however the stability studies have resulted in clear guidance for users of storage of the RM kit.
8.7 What is the kit intended for?
There are several possible applications for the food allergen RM kit. These include validation and verification studies of relevant (for example, relevant to the food allergens in the RM kit) analytical methods (ELISA, PCR, LC-MS/MS, LFD), and ELISA and PCR kits. Its applicability extends to new methods or detection kits, on-going competency assessments of existing detection kits and methods, and the competency of staff performing allergen analyses and of analytical service providers working in the food supply chain. The food allergen RM kit may be used directly to quality assure relevant non-routine analytical runs or to validate and verify in-house quality control materials for use in routine analytical runs.
Examples of usage include:
The raw materials can be used to
- generate kit calibrator extract solutions
- generate external check calibrator extract solutions
- spike various other matrices either by way of an extract but preferably by addition of the raw material itself to assess recovery in real life situations. It will be impossible to generate RMs for the required wide variety of matrices (for example, take-away curry type products) which are analysed by laboratories however the raw material may be spiked into such samples to provide matrix-specific recoveries.
The blank matrix can be used as
- a ‘no-template’ control to provide assurance of absence of in-lab allergen cross contamination (either environmentally, from personnel or in reagents)
- a material to assist method LoD calculation (as 3.3 times the standard deviation of a ‘blank’ dataset)
The incurred matric can be used to
- optimise analytical recovery from a chocolate-type matrix;
- inform risk assessors of the possible ‘true’ estimate of allergen in a questioned product.
- generate conversion factors from PCR copy number/copy number data to m/m data, and from peptides concentrations to protein and hence food concentrations (with appropriate definitions).
Allergen reference materials have been produced: hens’ egg white powder, skimmed cows’ milk powder, almond powder (full fat), hazelnut powder (partially defatted), and walnut powder (partially defatted) which are available as the appropriately characterised individual foods and added to a chocolate dessert paste to make a material at the clinically and industrially relevant incurred concentration of 10 mg kg-1 as the total protein. The homogeneity and stability of the materials were investigated and found to be fit for purpose.
The process of preparation and characterisation of the reference materials has been described. Traceability to the SI was achieved gravimetrically. The RM kit, which is on the market, has been confirmed within the scope of ISO 17034 accreditation. Statement of measurement have been published and assigned values compared with independently obtained data from two ELISA platforms.
The successful conclusion of this project does not, of course, solve all the problems in food allergen analysis and we make recommendations for further work to build on the firm foundations reported herein. These include:
- comparison of data from the reference materials on ELISA platforms other than the two exhibited in the project and two allergens (almond and walnut) were not extensively characterised in the project and the reference materials would benefit from further study, including in multiple laboratories.
- although the proteomics of some allergens in the kit have been reported there is scope for further work on the allergen profiles and on value assignment by liquid chromatography –tandem mass spectrometry (LC-MS/MS).
- value assignment for the nut ingredients by Polymerase Chain Reaction (PCR) DNA methods would add to extant data and achieve copy number to mass fraction conversion factors.
- homogeneity data in the reference materials are satisfactory but for hazelnut the data are more dispersed and it is not possible to distinguish the inherent variability of the ELISA from effects perhaps caused by the raw material particle size.The application of digital PCR, (dPCR) which offers much lower variance than ELISA methods and absolute single molecule quantification of DNA species without an external calibration curve, would give more precise information on the homogeneity of the reference materials.
- if dPCR homogeneity data retain the same dispersion driven by the particle size and mixing into the matrix exploration of cryogenic (-80C) milling would be useful. Assessment of the impact on protein structure of these low temperatures would also be interesting.
Funding is gratefully acknowledged from the UK Food Standards Agency (Project FS101206) and the UK Department for Business, Energy & Industrial Strategy.
Malvinder Singh, Kirstin Gray, Anuradha Balasundaram and Adrian Rogers are thanked for preparative and analytical input, Simon Cowen and Stephen L R Ellison are thanked for statistical evaluation off the data, Malcolm Burns is thanked for PCR DNA and bioinformatics support, Elena Sanchez is thanked for editorial input and William Webster is thanked for project financial management.
Appendix 1: Project Participants
A1.1 LGC
LGC is a global leader in the Life Science Tools sector, which serves customers across a number of end markets, including human healthcare, agri-food & the environment. LGC provides a comprehensive range of measurement tools, proficiency testing schemes, supply chain assurance standards and specialty genomics reagents underpinned by leading analytical and measurement science capabilities. Its scientific tools and solutions form an essential part of its customers’ quality assurance procedures and enable organisations to develop and commercialise new scientific products and advance research.
LGC’s 3,200+ employees include internationally-recognised scientists who are experts in their field. Headquartered in London, it operates out of 22 countries worldwide and is extensively accredited to quality standards such as GMP, GLP, ISO 13485, ISO 17034, ISO 17043, ISO/IEC 17025 and ISO 9001.
LGC has been home to the UK Government Chemist for more than 100 years and is the UK National Measurement Laboratory and Designated Institute for chemical and bio measurement. LGC has been privately-owned since 1996 and has diversified through internal investment and acquisition to be an international leader in its chosen markets.
For more information, please visit www.lgcgroup.com
About the National Measurement Laboratory
The National Measurement Laboratory (NML), hosted at LGC, delivers underpinning chemical and bio-measurement science for the UK and forms part of the UK National Measurement System (NMS). The NML is the Designated Institute for chemical and bio-measurement and supports the work of the Government Chemist, a unique statutory function that provides expert opinion and advice to Government.
Our research areas span the sectors of advanced therapeutics, diagnostics and safety & security and are delivered through the four core streams of measurement research, calibration facilities, reference materials, and training and consultancy. Our measurement capabilities comprise state-of-the-art mass spectrometry, PCR and cell characterisation of products and processes, with many of our testing and calibration services accredited to ISO/IEC 17025. We play a leading role internationally to develop best practice and standardise measurements across the world, in turn providing further confidence in the UK’s science and technology capabilities.
Through improved chemical and bio-measurements we support manufacture and trade, protect consumers, further skills development and enhance quality of life.
A1.2 University of Manchester Institute of Biotechnology
The strong interdisciplinary focus and mode of operation in the Manchester Institute of Biotechnology (MIB) enables our scientists and engineers to drive state-of-the-art biotechnology research through establishing new types of collaboration. The MIB’s approach is pluralistic, integrative and non-exclusive functioning flexibly as a partner to researchers across a broad spectrum of disciplines which are fluid and constantly evolving. Using advanced quantitative methods to explore the relationship between the macro behaviour of biological systems and the properties of their nanoscale components we see this understanding as a basis for developing new biotechnologies that will find applications in areas such as human health, the energy economy, food security, industrial transformations and the environment. A defining feature of our mission is the incorporation of rigorous, cutting-edge principles and technologies derived from the physical sciences, engineering, mathematics and computation into novel approaches to elucidating the molecular mechanisms that underpin living processes and systems. An extensive programme of technology development lies at the heart of our research programmes, driven by expertise from the quantitative sciences and engineering, allowing us to accelerate progress through the implementation of state-of-the-art enabling technologies.
A1.3 Romer Labs Ltd
Founded in Washington, MO, in 1982, Romer Labs became over the years a leading provider of diagnostic solutions for the agricultural, food and feed industry.
Today, Romer Labs offers a broad range of innovative diagnostics olutions covering mycotoxins, food pathogens, food allergens, gluten, GMO, veterinary drug residues, and other food contaminants. The portfolio includes:
- ELISA test kits - AgraQuant®
- Lateral flow devices - AgraStrip® and RapidChek®
- Reference materials - Biopure™
- Cleanup Columns - MycoSep®, MultiSep®, MycoSpin®, StarLine™
- Sampling mills
Furthermore, Romer Labs operate 4 accredited, full-service laboratories in Austria, UK, US and Singapore. Using cutting-edge technology in the fields of chromatography and immunological analysis, our labs offer services for the analysis of mycotoxins, food allergens, meat speciation, VDR and GMO.
Romer Labs is at the forefront of diagnostic technology and we are constantly expanding our product and service portfolio to meet your continuously evolving demands.
Our key objective at Romer Labs is to provide scientifically sound, high-quality products and an exceptional service, in line with our mission – Making the World’s Food Safer®.
Appendix 2: Refereed publications
The following refereed publication were prepared for publication during the project.
A2.1 Paper D1.1 - Pilolli et al.
Citation
Pilolli, R., Nitride, C., Gillard, N., Huet, A.C., van Poucke, C., de Loose, M., Tranquet, O., Larré, C., Adel-Patient, K., Bernard, H. and Mills, E.C., 2020. Critical review on proteotypic peptide marker tracing for six allergenic ingredients in incurred foods by mass spectrometry. Food Research International, 128, p.108747. DOI: 10.1016/j.foodres.2019.108747
Abstract
Peptide marker identification is one of the most important steps in the development of a mass spectrometry (MS) based method for allergen detection, since the robustness and sensitivity of the overall analytical method will strictly depend on the reliability of the proteotypic peptides tracing for each allergen. The European legislation in place issues the mandatory labelling of fourteen allergenic ingredients whenever used in different food formulations. Among these, six allergenic ingredients, namely milk, egg, peanut, soybean, hazelnut and almond, can be prioritized in light of their higher occurrence in food recalls for undeclared presence with serious risk decision. In this work, we described the results of a comprehensive evaluation of the current literature on MS-based allergen detection aiming at collecting all available information about proteins and peptide markers validated in independent studies for the six allergenic ingredients of interest. The main features of the targeted proteins were commented reviewing all details available about known isoforms and sequence homology particularly in plant derived allergens. Several critical aspects affecting peptide marker reliability were discussed and according to this evaluation a final short-list of candidate markers was compiled likely to be standardized and implemented in MS methods for allergen analysis.
A2.2 Paper D1.2 Adaba et al.
Citation
Rosemary Adaba, Chiara Nitride, Anuradha Balasundaram, Ernest Long, Ivona Bariceivic-Jones, Daniel Schaeffer, Jason Robothom, Gill Holcombe, Michael J Walker, and E. N. Clare Mills, 20xx, Characterisation of hazelnut, walnut and almond ingredient for use as reference materials for allergen analysis, (in preparation).
Abstract
Quantification of allergens in foods using different test methods can give rise to conflicting results, an issue that can be addressed by access to good quality incurred allergen reference materials which provide a mean of harmonising test results. Powdered ingredients were sourced from foods that frequently cause severe allergic reactions and/or give rise to product recalls and are representative of foods as they are commonly consumed. These included roasted peanut (Arachis hypogea), hazelnut, (Corylus avellana), walnut (Juglans regia) and almond (Prunus dulcis). The extraction of ingredients was optimised, and the allergen profiles characterised using a combination of immunoblotting using specific antibodies and discovery mass spectrometry using data dependent acquisition. Homologous allergens from the major plant food allergen families were identified across the plant species and shown to be present in an immunologically active form. These data demonstrate the suitability of the ingredients for the preparation of allergen reference materials.
A2.3 Paper D4.2 Holcombe et al.
Citation
Gill Holcombe, Michael J Walker, Malvinder Singh, Kirstin Gray, Simon Cowen, Stephen L R Ellison, Adrian Rogers, Anuradha Balasundaram, Malcolm Burns and E. N. Clare Mills, 20XX, Clinically and industrially relevant incurred reference materials to improve analysis of food allergens, milk, egg, almond, hazelnut and walnut, (paper submitted for publication).
Abstract
Measurement of critical food allergen protein concentrations against concentrations derived from quantitative allergen risk assessment has the potential to improve allergen risk management and precautionary allergen labelling. However, such measurement suffers well known problems. The availability of well characterised reference materials (RMs), called for by many, would allow comparability and harmonisation of measurements between methods and across different laboratories and help analysts provide meaningful information for risk assessors. We describe the preparation and characterisation of the first consensus informed industrially and clinically relevant multi-allergen matrix RM kit for five priority allergens. It consists of a medium analytical difficulty polyphenol-containing processed food chocolate paste matrix (a) devoid of allergens, and (b) incurred with five allergenic materials at the clinically relevant concentration of 10 mg kg-1 expressed as protein. The allergen raw materials: hens’ egg white powder, skimmed cows’ milk powder, almond powder (full fat), hazelnut powder (partially defatted), and walnut powder (partially defatted), are also available as RMs. The preparation, gravimetric traceability to the SI, homogeneity, short term (transportation) stability and long-term stability of the RM have been demonstrated and are described. Further, species characterisation by Sanger sequencing, and allergen profiling in the raw materials by a combination of immunoblotting and discovery mass spectrometry, demonstrate the suitability of the RM. The RM kit, which is on the market, has been confirmed within the scope of LGC’s ISO 17034 accreditation. Assigned values are compared with independently obtained data from two ELISA platforms and a statement of measurement has been published.
- Holcombe, G. 2020, Production of a Reference Material Kit for use in the detection and quantification of food allergens, LGC746-KT, LGC7421, LGC7422, LGC7424, LGC7425 and LGC7426.
- Poppy, G., 2016, Food Standards Agency, FSA, Chief Scientific Adviser’s Science Report Issue five: Food allergy and intolerance, PDF (Accessed 08.07.2019)
- Food Standards Agency, FSA, 2019, Summary of discussions at the FSA Board meeting on 19 June 2019, (Accessed 08.07.2019)
- Food Standards Agency, FSA, 2022, The FSA strategy for 2022 to 2027 (Accessed 03.02.2023)
- A. Muraro, T. Werfel, K. Hoffmann-Sommergruber, G. Roberts, K. Beyer, C. Bindslev-Jensen, V. Cardona, A. Dubois, G. du Toit, P. Eigenmann et al., on behalf of the EAACI Food Allergy and Anaphylaxis Guidelines Group, EAACI Food Allergy and Anaphylaxis Guidelines. Diagnosis and management of food allergy. Allergy 2014, 69, 1008–1025.
- S. Prescott and K. J. Allen, Food allergy: Riding the second wave of the allergy epidemic, Pediatr. Allergy Immunol., 2011, 22, 155–160.
- S. H. Sicherer and H .A. Sampson, Food allergy: epidemiology, pathogenesis, diagnosis, and treatment. J. Allergy Clin. Immunol., 2014, 133, 291-307.
- Nwaru, B. I., Hickstein, L., Panesar, S. S., Muraro, A., Werfel, T., Cardona, V., Dubois, A. E. J., Halken, S., Hoffmann-Sommergruber, K., Poulsen, L. K., Roberts, G., Van Ree, R., Vlieg-Boerstra, B. J., & Sheikh, A. (2014). The epidemiology of food allergy in Europe: a systematic review and meta-analysis. Allergy, 69(1), 62–75. https://doi.org/10.1111/all.12305
- T. Umasunthar, J. Leonardi‐Bee, M. Hodes, P. J. Turner, C. Gore, P. Habibi, J. O. Warner and R. J. Boyle. Incidence of fatal food anaphylaxis in people with food allergy: a systematic review and meta‐analysis, Clin. Exper. Allergy, 2013 43, 1333-1341.
- N.J. Avery, R. M. King, S. Knight and J. O’B. Hourihane, Assessment of quality of life in children with peanut allergy, Pediatr. Allergy Immunol., 2003, 14, 378–382.
- R. M. King, R. C. Knibb and J. O’B. Hourihane, Impact of peanut allergy on quality of life, stress and anxiety in the family. Allergy, 2009, 64, 461–468.
- C. Venter, I. Sommer, H. Moonesinghe, J. Grundy, G. Glasbey, V. Patil and T. Dean, Health‐Related Quality of Life in children with perceived and diagnosed food hypersensitivity. Pediatr. Allergy Immunol., 2015, 26, 126-132.
- B. Gibbison, A. Sheikh, P. McShane, C. Haddow and J. Soar, Anaphylaxis admissions to UK critical care units between 2005 and 2009, Anaesthesia, 2012, 67, 833-839.
- C. B. Madsen, S. Hattersley, K. J. Allen, K. Beyer, C‐H. Chan, S. B. Godefroy, R. Hodgson et al., Can we define a tolerable level of risk in food allergy? Report from a EuroPrevall/UK Food Standards Agency workshop, Clin. Exper. Allergy, 2012 42, 30-37.
- Lomer, M.C.E. (2015) Review article: the aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Alimentary Pharmacology and Therapeutics 41, 262–275.
- McIntosh, J., Flanagan, A., Madden, N., Mulcahy, M., Dargan, L., Walker, M. and Burns, D.T., 2011. Awareness of coeliac disease and the gluten status of ‘gluten‐free’food obtained on request in catering outlets in Ireland. International journal of food science & technology, 46(8), pp.1569-1574..
- Regulation (EC) No 178/2002 of the European Parliament and of the Council of 28 January 2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety. Off. J. Eur Commun., L31:1–24 (2002).
- H. Monks, M. H. Gowland, H. MacKenzie, M. Erlewyn-Lajeunesse, R. King, J. S. Lucas and G. Roberts, How do teenagers manage their food allergies? Clin. Exp. Allergy, 2010, 40, 1533–1540.
- JRC-ALLERGENS@ec.europa.eu
- G. O'Connor, M. Haponiuk (DG SANTÉ), F. Ulberth, Joint DG SANTÉ and DG JRC Workshop- Harmonisation of Approaches for informing EU allergen labelling legislation, JRC108259
- iFAAM publishable summary
- Benjamin C. Remington, Joseph Baumert, W. Marty Blom, Luca Bucchini, Neil Buck, René Crevel, Fleur De Mooij, Simon Flanagan, James Hindley, Bushra Javed, Despoina Angeliki Stavropoulou, Myrthe W. van den Dungen, Marjan van Ravenhorst, Si Wang, Michael Walker, 2022, Allergen quantitative risk assessment within food operations: Concepts towards development of practical guidance based on an ILSI Europe workshop, Food Control, 138, 108917, https://doi.org/10.1016/j.foodcont.2022.108917.
- Benjamin Remington, Joseph Baumert, Marty Blom, Luca Bucchini, Neil Buck, René Crevel, Fleur De Mooij, Simon Flanagan, Angeliki Stavropoulou, Myrthe van den Dungen, Marjan van Ravenhorst, Michael Walker and Si Wang, 2022, Practical Guidance on the Application of Food Allergen Quantitative Risk Assessment (QRA), ILSI Europe Report Series, 13/06/2022, https://doi.org/10.5281/zenodo.6651934 (https://ilsi.eu/publication/practical-guidance-on-the-application-of-fo… )
- Taylor SL, Baumert JL, Kruizinga AG, Remington BC, Crevel RW, Brooke-Taylor S, Allen KJ, Allergen Bureau of Australia & New Zealand, Houben G. (2014) Establishment of Reference Doses for residues of allergenic foods: report of the VITAL Expert Panel. Food Chem Toxicol.; 63:9-17
- Codex Alimentarius Commission. CXC 80-2020, code of practice on food allergen management for food business operators. FAO, Rome (Accessed 11.02.2023)
- Summary report of the Ad hoc Joint FAO/WHO Expert Consultation on Risk Assessment of Food Allergens. Part 1: Review and validation of Codex priority allergen list through risk assessment (available at http://www.fao.org/3/cb4653en/cb4653en.pdf and https://cdn.who.int/media/docs/default-source/food-safety/jemra/1st-all…) (Accessed 11.02.2023)
- FAO and WHO. 2022. Risk Assessment of Food Allergens. Part 1 – Review and validation of Codex Alimentarius priority allergen list through risk assessment. Meeting Report. Food Safety and Quality Series No. 14. Rome. https://doi.org/10.4060/cb9070en (Accessed 11.02.2023)
- Summary report of the Ad hoc Joint FAO/WHO Expert Consultation on Risk Assessment of Food Allergens. Part 2: Review and establish threshold levels in foods of the priority allergens (Accessed 11.02.2023)
- WHO/FAO Risk Assessment Of Food Allergens Part 2: Review and Establish Threshold Levels in Food for The Priority Allergens, 2022, (Accessed 11.02.2023)
- Summary report of the Ad hoc Joint FAO/WHO Expert Consultation on Risk Assessment of Food Allergens. Part 3: Review and establish precautionary labelling in foods of the priority allergens. (Accessed 11.02.2023)
- Ad hoc Joint FAO/WHO Expert Consultation on Risk Assessment of Food Allergens Part 4: Review and establish exemptions for the food allergens FAO HQ, Rome, Italy: 14 - 18 November 2022 Summary and Conclusions Issued in January 2023 (Accessed 11.02.2023)
- Walker, Michael John, Duncan Thorburn Burns, Chris Elliott, M. Hazel Gowland, and E N Clare Mills, 2016, ‘Flawed food allergen analysis–health and supply chain risks and a proposed framework to address urgent analytical needs’, Analyst, 141, 24 – 35.
- Rzychon, M., Brohée, M., Cordeiro, F., Haraszi, R., Ulberth, F. and O'Connor, G., 2017. The feasibility of harmonizing gluten ELISA measurements. Food chemistry, 234, pp.144-154.
- Taylor, S.L., Nordlee, J.A., Niemann, L.M. and Lambrecht, D.M., 2009. Allergen immunoassays—considerations for use of naturally incurred standards. Analytical and Bioanalytical Chemistry, 395(1), pp.83-92.
- Mills, E.C., Adel-Patient, K., Bernard, H., De Loose, M., Gillard, N., Huet, A.C., Larré, C., Nitride, C., Pilolli, R., Tranquet, O. and Pouke, C.V., 2019. Detection and quantification of allergens in foods and minimum eliciting doses in food-allergic individuals (ThRAll). Journal of AOAC International, 102(5), pp.1346-1353.
- Huet, A.C., Paulus, M., Henrottin, J., Brossard, C., Tranquet, O., Bernard, H., Pilolli, R., Nitride, C., Larré, C., Adel-Patient, K. and Monaci, L., 2022. Development of incurred chocolate bars and broth powder with six fully characterised food allergens as test materials for food allergen analysis. Analytical and Bioanalytical Chemistry, 414(8), pp.2553-2570.
- Huet, A.C., Paulus, M., Henrottin, J., Brossard, C., Tranquet, O., Bernard, H., Pilolli, R., Nitride, C., Larré, C., Adel-Patient, K. and Monaci, L., 2022. Development of incurred chocolate bars and broth powder with six fully characterised food allergens as test materials for food allergen analysis. Analytical and Bioanalytical Chemistry, 414(8), pp.2553-2570.
- Martinez-Esteso, Maria José, Gavin O’Connor, Jørgen Nørgaard, Andreas Breidbach, Marcel Brohée, Elena Cubero-Leon, Chiara Nitride, Piotr Robouch, and Hendrik Emons. 2020. “A Reference Method for Determining the Total Allergenic Protein Content in a Processed Food: The Case of Milk in Cookies as Proof of Concept.” Analytical and Bioanalytical Chemistry 412 (30): 8249–67. https://doi.org/10.1007/s00216-020-02959-0.
- Nitride, Chiara, Jørgen Nørgaard, Jone Omar, Hendrik Emons, María José Martínez Esteso, and Gavin O’Connor. 2019. “An Assessment of the Impact of Extraction and Digestion Protocols on Multiplexed Targeted Protein Quantification by Mass Spectrometry for Egg and Milk Allergens.” Analytical and Bioanalytical Chemistry 411 (16): 3463–75. https://doi.org/10.1007/s00216-019-01816-z.
- Cordeiro, Fernando, Elena Cubero-Leon, Jørgen Nørgaard, Maria José Martinez-Esteso, Marcel Brohée, Andreas Breidbach, Aneta Cizek-Stroh, Gavin O’Connor, Piotr Robouch, and Hendrik Emons. 2021. “Total Cow’s Milk Protein in Cookies: The First Interlaboratory Comparison with a Well-Defined Measurand Fit for Food Allergen Risk Assessment.” Accreditation and Quality Assurance 26 (3): 177–81. https://doi.org/10.1007/s00769-021-01470-y.
- Breidbach, Andreas, Jørgen Vinther Nørgaard, Elena Cubero-Leon, and Maria Jose Martinez Esteso. 2022. “Assignment of a Reference Value of Total Cow’s Milk Protein Content in Baked Cookies Used in an Interlaboratory Comparison.” Foods 11 (6): 869. https://doi.org/10.3390/foods11060869.
- Sicherer, S.H. and Sampson, H.A., 2018. Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. Journal of Allergy and Clinical Immunology, 141(1), pp.41-58.
- Bilaver, L.A., Chadha, A.S., Doshi, P., O'Dwyer, L. and Gupta, R.S., 2019. Economic burden of food allergy: a systematic review. Annals of Allergy, Asthma & Immunology, 122(4), pp.373-380.
- M.H. Gowland, M.J. Walker, 2014, Food Allergy, a summary of 8 cases in the UK criminal and civil courts: effective last resort for vulnerable consumers?, J. Sci. Food Agric., 95: 1979–1990
- In The West London Coroners Court, Before Dr Sean Cummings, HM Assistant Coroner London (Western Area), The Inquest Touching the Death of Natasha Ednan-Laperouse
- Mohammed Abdul Kuddus v R., Court of Appeal (Criminal Division), [2019] EWCA Crim 837 (16 May 2019)
- Cummings, S. Natasha Ednan-Laperouse. London (West). Report to prevent future deaths Date: 2018 (Accessed 13.07.2019)
- Audrey DunnGalvin, Graham Roberts, Sabine Schnadt, Siân Astley, Moira Austin, W. Marty Blom, Joseph Baumert, Chun‐Han Chan, René W.R. Crevel, Kate E C Grimshaw, Astrid G. Kruizinga, Lynne Regent, Stephen Taylor, Michael Walker, Clare Mills, 2019. Evidence based approaches to the application of Precautionary Allergen Labelling: Report from two iFAAM workshops. Clinical & Experimental Allergy, 20 July 2019, https://doi.org/10.1111/cea.13464
- S. L. Taylor, J. L. Baumert, A. G. Kruizinga, B. C. Remington, R. W. R. Crevel, S. Brooke-Taylor, K. J. Allen, The Allergen Bureau of Australia, and G. Houben, Establishment of reference doses for residues of allergenic foods: report of the VITAL expert panel, Food Chem. Toxicol., 2014, 63, 9-17.
- Mattarozzi, M. & Careri, M. 2019, The role of incurred materials in method development and validation to account for food processing effects in food allergen analysis, Anal Bioanal Chem 411: 4465
- Johnson, P.E. and Downs, M., 2019. From Signal to Analytical Reporting for Allergen Detection by Mass Spectrometry. Journal of AOAC International, 102(4) July-August 2019, https://doi.org/10.5740/jaoacint.19-0053
- Phil E. Johnson, Neil M. Rigby, Jack R. Dainty, Alan R. Mackie, Ulrike U. Immer, Adrian Rogers, Pauline Titchener, Masahiro Shoji, Anne Ryan, Luis Mata, Helen Brown, Thomas Holzhauser, Valery Dumont, Jill A. Wykes, Michael Walker, Jon Griffin, Jane White, Glenn Taylor, Bert Popping, René Crevel, Sonia Miguel, Petra Lutter, Ferdelie Gaskin, Terry B. Koerner, Dean Clarke, Robin Sherlock, Andrew Flanagan, Chun-Han Chan, E.N. Clare Mills,2014, A multi-laboratory evaluation of a clinically-validated incurred quality control material for analysis of allergens in food, Food Chemistry, 148, 30-36
- Gill Holcombe, Michael J Walker, Malvinder Singh, Stephen L R Ellison, Joanna Topping, Adrian Rogers, Phil E Johnson, Anuradha Balasundaram and E. N. Clare Mills, 2019, Clinically relevant incurred reference materials for the quality control of analysis of peanut allergen in food – preparation and availability, IN PREPARATION
- Regulation (EU) No. 1169/2011 of the European Parliament & of the Council of 25 October 2011 on the provision of food information to consumers, OJ L 304, 22.11.2011, 18–63
Revision log
Published: 30 May 2023
Last updated: 30 May 2024