Development of reference materials: Food hypersensitivity
Food hypersensitivity reactions to food have resulted in considerable morbidity and reached epidemic proportions in the industrialized world.
4.1 Background
Food hypersensitivity, for example, adverse immunologic (IgE and non-IgE mediated) reactions to food, have resulted in considerable morbidity [5] and reached epidemic proportions in the industrialized world [6], [7] affecting 5 – 6% of young children and 1 -2% of adults [8]. Anaphylaxis, a rapid onset multi-organ system allergic reaction can cause fatalities. The risk of such deaths, though comparatively rare [9], contributes to well-documented detriment to the quality of life for allergic consumers and their families [10], [11], [12].
There are burdens on health care [13], on businesses (food recalls, for example) and regulators [14] and in less developed countries where, owing to poor labelling and awareness, significant challenges may exist. Current reputed cures for food allergies remain experimental and lifelong avoidance of the eliciting food(s) is required. Autoimmune conditions such as coeliac disease and food intolerance [15] also impose significant burdens [16] and strict avoidance of the eliciting foods is usually necessary.
4.2 Food allergen regulation
UK and EU legislation requires food manufacturers to list the ingredients of a food product on its packaging. If one of the 14 regulated allergens (Table 3) (chosen based on the prevalence of adverse reactions amongst consumers in the 27 EU member states) is used as an ingredient or processing aid it must be highlighted within this ingredients list.
The approach for unintended allergens is different: Unintended allergens may enter into the food supply chain at any time during harvest, storage, handling, and/or packaging as consequence of the cross-contact or carry over within processing lines. This represents a food safety hazard. Food business operators should implement a risk assessment in order to establish whether a hazard is likely to occur, and seek to idea; eliminate this risk, or, where this is not feasible, reduce the risk of contamination to acceptable levels, below which no, or only the most sensitive, allergenic subjects might react.
Precautionary Allergen Labelling should be used when there is an unavoidable risk of allergen cross-contact that cannot be sufficiently controlled. Uncertainty around the risk assessing allergen cross-contact has resulted in the untargeted use of Precautionary Advisory Labelling (PAL) - for example, ‘may contain...’. This has caused subsequent confusion of the exact meaning of such warnings amongst consumers [17], [18].
Table 3: Allergens covered by EU labelling law, Annex II Regulation 1169/2011 (UK Retained Regulation (EU) 1169/2011)
| Annex II Entry | Examples |
|---|---|
| Cereals containing gluten and product thereof | Wheat, Rye, Barley, Oats |
| Crustaceans and products thereof | Shrimp/prawn, Crab, Lobster, Crayfish |
| Eggs and products thereof | - |
| Fish and products thereof | - |
| Peanuts and products thereof | - |
| Soybeans and products thereof | - |
| Milk and products thereof | Skimmed milk powder, Cheese etc. |
| Nuts and nut products namely | Almond, Hazelnut, Walnut, Cashew, Pecan, Brazil, Pistachio, Macadamia |
| Celery and products thereof | - |
| Mustard and products thereof | - |
| Sesame seed and products thereof | - |
| Sulphur dioxide/sulphites | - |
| Lupin and products thereof | - |
| Molluscs and products thereof | Mussels, Scallops, Cockles, Oyster, Clam |
There are limited exceptions cited in Annex II of ingredients that do not contain sufficient allergenic protein to elicit a reaction.
4.3 Allergen risk assessment
4.3.1 DG Santé/JRC workshop
In 2016, DG Santé/JRC organised a joint workshop aiming ‘to identify the sequence of steps required for framing the current use of precautionary allergen information and its enforcement across the EU and UK [19]. The major points of the discussion were: (1) legislative and allergy sufferers’ requirements; (2) risk based approaches to allergen management; (3) the role of analysis in enforcing legislation. The outcomes of this discussion will have a significant impact on analytical measurement requirements. As stated in the available feedback report:
- 'Possible agreement on analytical marker(s) and their conversion to a common reporting unit should be encouraged.
- the most appropriate reporting unit for reporting analytical results is mg total allergenic protein ingredient per kg food.
Establishing an expert group to facilitate the progression of all allergenic foods to report in this manner was thought beneficial. This group should consider work done by CEN and other standardisation bodies in the area.
4.3.2 Risk assessment toolkit development
Risk assessment toolkits were developed by the Allergen Bureau Voluntary Incidental Trace Allergen Labelling, VITAL® and the Integrated Approaches to Food Allergen and Allergy Management (iFAAM) consortium to manage food allergen risk [20]. The practical application of quantitative allergen risk assessment has been described by an ILSI-Europe Expert Group [21], [22]. The toolkits use action levels to determine if a corrective action is required. The action level is a combination of the estimated eliciting dose for the different allergens (ED) and the amount of food consumed in a single eating occasion. The eliciting dose is the predicted amount of allergenic food, expressed as mg of total protein that can provoke an allergic reaction in a given percent of the allergic population. It is extrapolated from dose-distribution relationships that are developed using oral food clinical challenge data. Table 4 and equation 1 show the VITAL® 2.0 and VITAL® 3.0 estimated reference doses and the related critical concentrations (mg kg-1 as protein) of the allergen in the food consumed above which an unacceptable risk is to be expected (action level) calculated for a specific amount of food consumed [23].
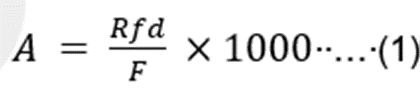
Where:
A: the critical concentration (mg kg-1 as protein) of the allergen in the food consumed
Rfd: the reference dose, in mg as protein as an eliciting dose at a specific population fraction
F : a representative amount of food consumed as g, (x1000 converts to mg kg-1 )
4.3.3 Codex Alimentarius code of practice
In 2020 Codex Alimentarius published a ‘Code of Practice on Food Allergen Management for Business Operators’/ [24]. As part of the ongoing review of the General Standard for the Labelling of Prepackaged Foods Codex also asked the Food and Agriculture Organisation of the United Nations (FAO) and the World Health Organisation (WHO) to look at food allergen risk assessment. A series of four ad hoc Joint FAO/WHO Expert Consultations was set up.
The first reviewed criteria for listing as priority allergens and affirmed the protein based allergens in the Codex list with the addition of sesame. It was recommended that pulses, insects and other foods such as kiwi fruits be included in a “watch list”. The committee also considered analytical capabilities and recommended the development of method performance criteria, more extensive provision of accessible reference materials, better understanding of assay performance and improvements in sampling and curation of samples from originator to laboratory [25]. A full report is available [26].
The second expert consultation discussed ‘thresholds’ for the priority allergens. The experts considered evidence that symptoms up to ED05 doses are mild or moderate. Up to 5 percent of reactions at both ED01 and ED05 could be classed as anaphylaxis, although none were severe. No fatal reactions at or below ED01 and ED05 were reported in the literature. The committee recommended reference doses at ED05 of 1 milligram of the relevant total protein for almond, cashew, pecan, pistachio and walnut, 2 milligrams for peanut, egg, sesame and cows’ milk protein, 3 milligrams for hazelnut protein and for wheat and fish the ED05 reference dose of 5 milligrams. The recommended ED05 for crustacea protein is much higher at 200 mg total protein. A summary [27] and a full report are available[28].
The third consultation discussed precautionary allergen labelling, recognising its difficulties. Their recommendations including that fit for purpose analytical methods are required, including for example matrix-matched assay validation with a limit of quantification at least 3-fold below the action level for the specific food, and reporting in units of milligrams of total protein from the allergenic source per kilogram of food [29].
The fourth consultation considered whether certain highly refined foods and ingredients that are derived from any of the foods on the list of priority allergenic foods could be exempted from mandatory labelling declaration. The consultation aimed to develop a process for the consideration of future such exemptions [30].
Table 4 VITAL® 2.0 and VITAL® 3.0 reference values and calculated action levels to portion size.
Action level (mg kg-1 as protein) based on VITAL 3.0, by way of illustration.
| Allergen | Reference dose (mg of protein): VITAL® 2.0 (V) and/or EACCI (E) *ED01 or #ED05 | Reference dose (mg of protein): VITAL® 3.0 (2019) All ED01 | Examples of amount of food consumed 5g | Examples of amount of food consumed 50g | Examples of amount of food consumed 250g |
|---|---|---|---|---|---|
| Peanut | 0.2* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Cow's milk | 0.1* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Egg | 0.03* (V,E) | 0.2 | 40 | 4 | 0.8 |
| Hazelnut | 0.1* (V,E) | 0.1 | 20 | 2 | 0.4 |
| Soya | 1.0* (V,E) | 0.5 | 100 | 10 | 2.0 |
| Wheat | 1.0# (V,E) | 0.7 | 140 | 14 | 2.8 |
| Cashew | 0.10# (V,E) | 0.05 | 10 | 1.0 | 0.2 |
| Mustard | 0.05# (V,E) | 0.05 | 10 | 1.0 | 0.2 |
| Lupin | 4# (V,E) | 2.6 | 520 | 52 | 10 |
| Sesame seed | 0.2# (V,E) | 0.1 | 20 | 2 | 0.4 |
| Shrimp | 10# (V,E) | 25 | 5000 | 500 | 100 |
| Fish | 0.1# (V,E) | 1.3 | 260 | 26 | 5 |
Note 1: The amounts shown here are only for illustration purposes. For guidance on how to estimate the amount of food consumed see ILSI-Europe, Practical Guidance on the Application of Food Allergen Quantitative Risk Assessment, Section 5.3: Guidance on food intake data for allergen risk assessment.
Note 2: In the last three columns of Table 4 only the appropriate number of significant figures have been retained.
4.4 Food allergen analysis
Reliable analytical measurement of allergens is required for many reasons [32] including to support the validation and verification of risk assessment plans in food industrial plants. This will allow food business operators to obtain quantitative values, expressed as mg of total allergenic protein per kg of food product, to be used in the process of decision-making.
4.4.1 Enzyme-Linked Immunosorbent Assay (ELISA)
The food industry and enforcement scientists (Public Analysts) largely employ Enzyme-Linked Immunosorbent Assay (ELISA) for routine assessment of food allergen concentrations. The technique is relatively easy to use, specific, not overly time consuming, and exhibits suitably low limits of detection and quantification for many allergens. However there are limitations. The assay can detect only one allergen per test and responses can be affected by nonspecific antibody reactivity and the non-reversible modification of the targeted protein(s) during food processing can mask the epitope to the antibody hence the risk of false positive or negative results is high. The lack of standardization can generate conflicting results among ELISA kit manufacturers. In proficiency testing multimodal datasets for allergen ELISAs are common and different assigned values have to be generated for the different kits used. Cross reactivity’s to foods other than the intended target have been reported for what were assumed to be specific ELISAs.
4.4.2 Polymerase Chain Reaction (PCR)
Polymerase Chain Reaction (PCR) assays are also applied in allergen analysis. They do not directly measure the hazard (protein), and are probative of the source species DNA rather than the allergen protein. They are especially valuable for those foods for which no ELISA based test is available and often have advantages in terms of sensitivity and specificity. In addition, as an analyte DNA is generally more stable than proteins although DNA degradation does occur by extremes of, for example, temperature and pH. PCR is essentially qualitative at present. Quantification based on DNA copy number can be derived from cycle thresholds but requires reference materials to construct a calibration curve (although digital PCR may circumvent this difficulty). However at present it is not easy to convert a quantification based on copy number to a weight/weight basis.
4.4.3 Mass Spectrometry (MS)
Mass spectrometry (MS) platforms are becoming the gold standard technologies for protein characterisation and quantification. Shotgun and targeted protein identification and quantification by mass spectrometry have been successfully applied to the analysis of allergens in processed food matrices.
Mass spectrometry allows a bottom-up analysis using harsher extraction protocols than can be used with ELISA (where it is essential to preserve the intact and native structure of the protein). Under MS, crude extracted protein can be broken down into its constituent peptides using enzymatic digestion.
The shotgun approach provides information on protein/peptide structure and on chemical/thermal modifications occurring during food processing. The targeted approach, by monitoring specific peptide markers as surrogates of given allergenic proteins, is capable of measuring many analytes in parallel, resolving very complex proteomes and dealing with protein abundances different by four to five orders of magnitude, while maintaining high mass accuracy. However, the technique requires a high level of expertise and costly equipment. More importantly, the preparation of the analytes for mass spectrometry analysis, including extraction and digestion of the proteins, remains challenging.
4.4.4 Overview
In summary, current industrial practice is heavily reliant on ELISA, and the method has brought many benefits in allergen risk management. However it exhibits some deficiencies which may jeopardise present and future risk management strategies. Alternative methods of allergen analysis are promising, but are also not without their challenges. The promise of MS or PCR may be lost if underpinning work suggested herein is not carried out. Structural changes in the target molecules induced by food processing or sample extraction may prevent detection by all methods.
Revision log
Published: 30 May 2023
Last updated: 30 May 2024